Nanotechnology And Its Role In Automotive Surface Care

What is nanotechnology?
There are a lot of car care products on the market today which claim to employ ‘nanotechnology’, stating nano or nanotech products last longer and work better. But what is nanotechnology and what role does it play in car care?
Defined simply, nanotechnology is the manipulation of matter at subatomic level (smaller than an atom). ‘Nano’, like ‘milli’, is actually a unit of measurement.
Nanotechnology is the science that deals with structures with at least one dimension measuring between 1-100 nanometres (1nm = 1 billionth of a metre. – National Institute for Nanotechnology (Edmonton, Canada)
To put it in context – 1mm is the smallest division on a ruler, the thickness of about 10 sheets of paper. A micrometre is 1000 times smaller than a millimetre. A nanometre is 1000 times smaller still. No surprise then that ‘nano’ comes from the Greek word for ‘dwarf’.
Materials structured on the nanoscale can have properties quite different from those of the same substance in a bigger form. This is because the ratio of the surface area of a material relevant to its volume plays a crucial role in its performance. Nanoscale materials have a greater proportion of their atoms on the surface than bulk materials and this affects many properties including strength, melting point (or heat resistance) and hardness. In addition some compounds when reduced to the nanoscale become optically clear (i.e. they do not scatter visible light) but retain their UV absorption properties. All of which are highly desirable properties for sealants designed to protect automotive surfaces.

‘Smart’ materials have the ability to interact with their environment. ‘Smart’ battery chargers react to drops in battery levels, smart coatings constructed on a nanoscale react to prevent ingress of water or dirt. Indeed in 2003 a popular science book on nanotechnology proposed the idea that:
Suppose that stain preventers could be incorporated permanently into clothing so that spills would no longer mean a trip to the dry cleaner. Suppose that automobile windscreens did not get wet so that no ice would ever form on them and rain could not impede visibility – (Ratner & Ratner, 2003)

Hydrophilic, Hydrophobic or Superhydrophobic?
Metals are inherently negatively charged and therefore hydrophilic (water loving). This means that water droplets (and the contaminants within them) falling on to the unprotected surfaces of your car, inherently form low contact angles, spread out and wet the surface thus leaving their contents behind.
Hydrophobicity is an important factor in any automotive sealant (be it a natural carnauba wax or synthetic). Unavoidably, rain carries with it industrial, chemical and environmental contaminants which quickly adhere to automotive surfaces. A hydrophobic barrier encourages the water to bead and roll away taking with it the contamination, rather than settling onto the underlying surface. This is often referred to as self-cleaning and makes washing the vehicle easier as the surfaces are less contaminated. This in turn reduces the risk of introducing swirls and scratches.
When a droplet of water rests on a surface, there is a contact angle measured at the edge of the droplet. (figure 1. below)
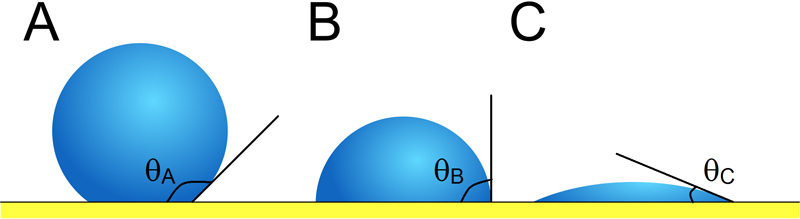
Typically, if a surface has a water contact angle of less than (<) 90 degrees (A), it is described as hydrophilic. Surfaces with a water contact angle between 90 and 150 degrees (B) are classified as hydrophobic and are distinguishable by water forming into droplets (beading). The more spherical the droplets the higher the water contact angle and the greater the hydrophobicity (water repelling properties).
When the water contact angle becomes greater than (>) 150 degrees (C) the surface is deemed as being superhydrophobic (commonly termed the ‘Lotus Effect’ after the leaves of the lotus plant). Superhydrophobic surfaces are highly hydrophobic, i.e., extremely difficult to wet and have a roll-off angle that is typically less than 10 degrees.

It is commonly accepted that superhydrophobic surfaces are achieved by increasing the roughness of an intrinsically hydrophobic substrate in order to maintain the highest water contact angle. Currently the science of nanotechnology has not advanced enough to produce stable, optically clear superhydrophobic coatings suitable for automotive sealant applications where both stability and clarity is a prerequisite.
Figure 2: Showing the surface structure required to achieve superhydrophobicity.

We are, however, seeing major leaps forward in automotive surface nanotechnology with new sealants from companies such as innovative Japanese manufacturer Kamikaze Collection and UK-based Gtechniq leading the way.
Products such as Kamikaze Collection ISM Hydrocarbon Coating and Gtechniq EXO v4 utilise a sol-gel chemical process to put down a self-organising interlaced molecular net of silica and other nano-scale components to form an extremely thin (< 0.1 micron) transparent glass-like coating that chemically bonds to the paints clear coat.
Whilst this new generation of products do not quite match the superhydrophobic attributes demonstrated by Mother Nature’s Lotus leaf, they do possess exceptional hydrophobicity (water resistance) and oleophobicity (oil repellance), the combination of which assists these coatings in obtaining their exceptional self-cleaning properties.
As momentum in this sector gathers pace, it won’t be too long before scientists produce the first genuine superhydrophobic coating, until then watch this space!

The full range of UF products can be found here.
Visit Ultimate Finish for the best car care and detailing products from around the globe.
Have a question on any of our products? Email sales@theultimatefinish.co.uk or call 01474 360 360.
Our team is on hand, Monday to Friday, 8am-5pm.